AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Compton scattering in x ray3/2/2024 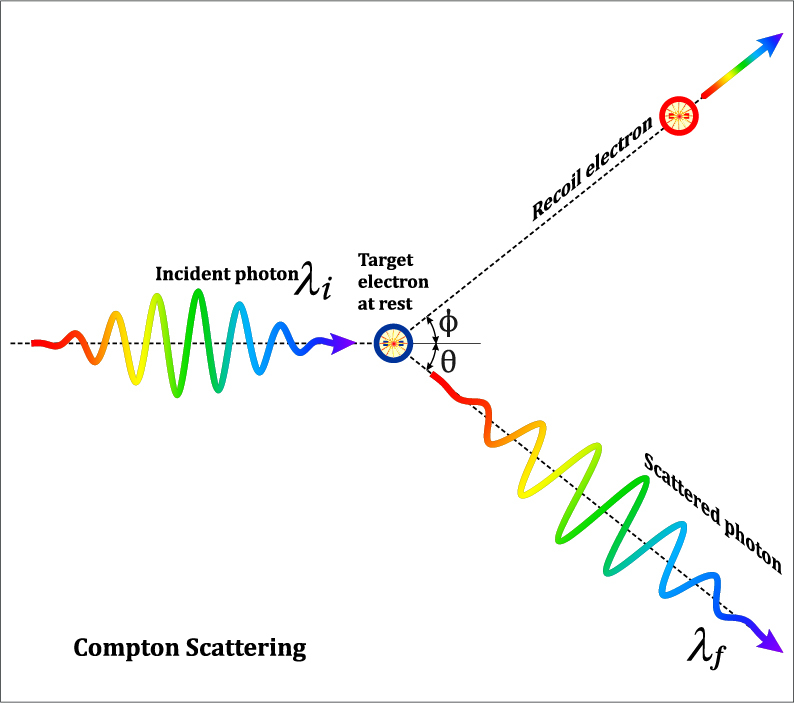
An electron from a more outer shell, with a lower binding energy, will fill this vacancy. As a result of a photoelectric interaction, atoms are ionized with a vacancy of an inner shell electron. For example, photons whose energies exceed the K-shell binding energies will most likely result in photoelectric interactions with the K-shell electrons. For a photoelectric effect to occur, the incident photon energy must be equal to or greater than the binding energy of the ejected electron. Illustration of the photoelectric effect.For example, an iodine atom which has a K-edge of 33.2 keV is six times more likely to have a photoelectric interaction with a 33.2 keV X-ray photon than with a 33.1 keV photon ( Figure 2.4). Every element has one or a few sharp “absorption edges” in which the probability of a photoelectric interaction dramatically increases for photons of energies just above the absorption edge relative to energies just below the edge. As the incident photon energy increases to just above the binding energy of the K shell, the absorption of the photon markedly increases. However, there are sharp discontinuities to be observed at certain photon energies. Also, the probability of photoelectric absorption generally decreases with the increase in photon energy. This is why barium ( Z = 56) and iodine ( Z = 53), which have high atomic numbers, are used as contrast agents in X-ray imaging. Therefore, as the atomic number increases, the photoelectric absorption effect becomes more pronounced. The probability of photoelectric absorption, by approximation, is considered proportional to Z 3/E 3, where Z is the atomic number and E is the energy of the incident photon.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
